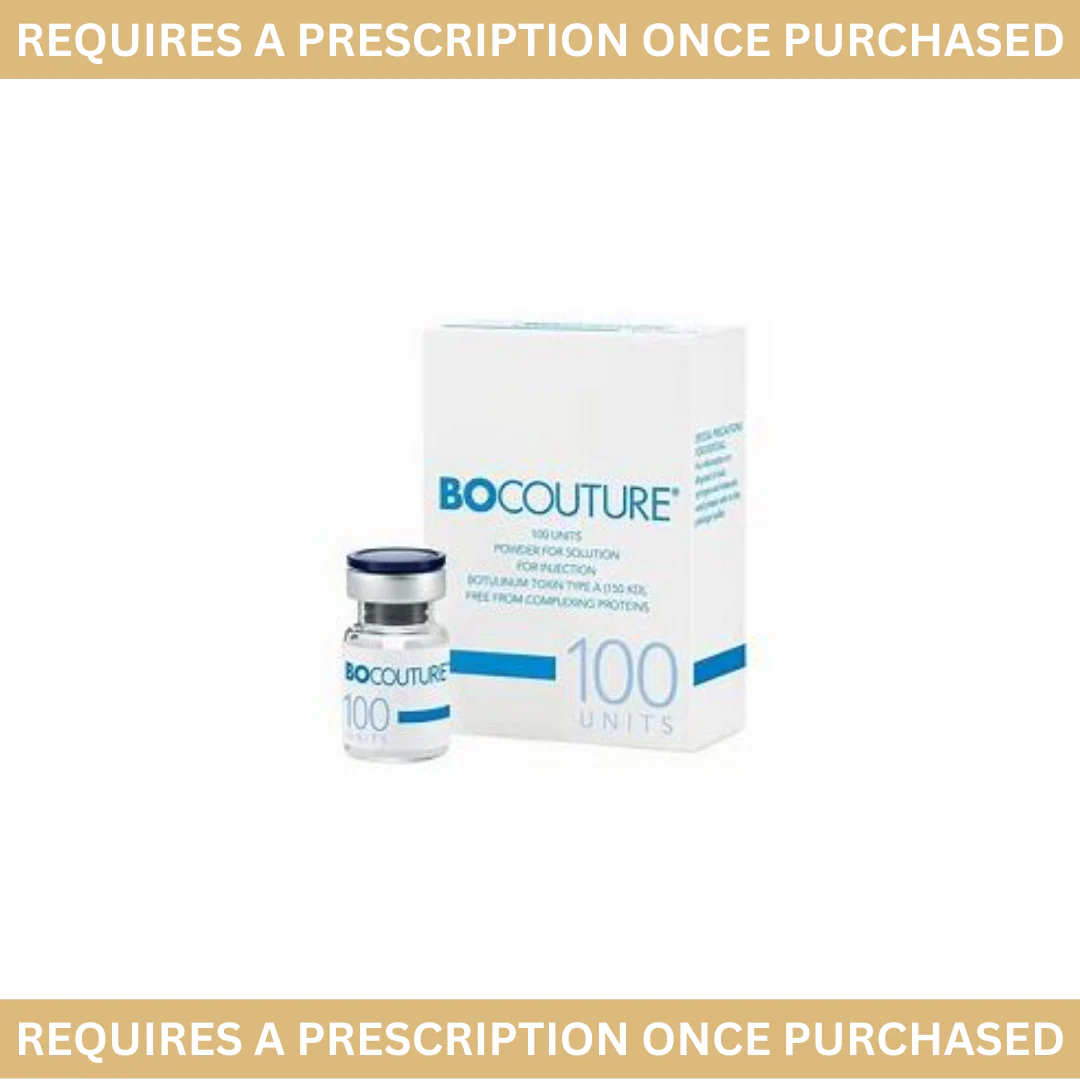
Bocouture
Bocouture is a prescription-only medicine indicated for the temporary improvement in the appearance of moderate to severe vertical lines between the eyebrows. Suitable for the treatment of glabellar frown lines, periorbital lines crow’s feet lines and horizontal forehead lines in adult patients under 65 years.
Summary:
- Prescription-only medicine
- Toxin indicated for the temporary improvement of lines and wrinkles
- Suitable for the forehead and periorbital areas
- Free from complexing proteins
Mode of action:
- Botulinum neurotoxin type A blocks cholinergic transmission at the neuromuscular junction by inhibiting the release of acetylcholine.
- The nerve terminals of the neuromuscular junction no longer respond to nerve impulses, and secretion of the neurotransmitter at the motor endplates is prevented.
- The action results in stopping the muscular contraction of the targeted muscles.
Contraindications of Bocouture:
- Hypersensitivity to the active substance or to any of the excipients.
- Generalised disorders of muscle activity (e.g. myasthenia gravis, Lambert- Eaton syndrome).
- Infection or inflammation at the proposed injection site.
Who should not use Bocouture?
- The injection of Bocouture is not recommended for patients with a history of aspiration or dysphagia.
- Patients suffering from amyotrophic lateral sclerosis
- Patients with other diseases which result in peripheral neuromuscular dysfunction
- In targeted muscles which display pronounced weakness or atrophy
EMC: https://www.medicines.org.uk/emc/product/7418/smpc
Patient Information Leaflet: https://www.medicines.org.uk/emc/files/pil.7418.pdf
*This item requires a prescription from a registered UK prescriber before it can be dispatched*
More Images
Bocouture
Bocouture is a prescription-only medicine indicated for the temporary improvement in the appearance of moderate to severe vertical lines between the eyebrows. Suitable for the treatment of glabellar frown lines, periorbital lines crow’s feet lines and horizontal forehead lines in adult patients under 65 years.
Summary:
- Prescription-only medicine
- Toxin indicated for the temporary improvement of lines and wrinkles
- Suitable for the forehead and periorbital areas
- Free from complexing proteins
Mode of action:
- Botulinum neurotoxin type A blocks cholinergic transmission at the neuromuscular junction by inhibiting the release of acetylcholine.
- The nerve terminals of the neuromuscular junction no longer respond to nerve impulses, and secretion of the neurotransmitter at the motor endplates is prevented.
- The action results in stopping the muscular contraction of the targeted muscles.
Contraindications of Bocouture:
- Hypersensitivity to the active substance or to any of the excipients.
- Generalised disorders of muscle activity (e.g. myasthenia gravis, Lambert- Eaton syndrome).
- Infection or inflammation at the proposed injection site.
Who should not use Bocouture?
- The injection of Bocouture is not recommended for patients with a history of aspiration or dysphagia.
- Patients suffering from amyotrophic lateral sclerosis
- Patients with other diseases which result in peripheral neuromuscular dysfunction
- In targeted muscles which display pronounced weakness or atrophy
EMC: https://www.medicines.org.uk/emc/product/7418/smpc
Patient Information Leaflet: https://www.medicines.org.uk/emc/files/pil.7418.pdf
*This item requires a prescription from a registered UK prescriber before it can be dispatched*
Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
Bocouture is a prescription-only medicine indicated for the temporary improvement in the appearance of moderate to severe vertical lines between the eyebrows. Suitable for the treatment of glabellar frown lines, periorbital lines crow’s feet lines and horizontal forehead lines in adult patients under 65 years.
Summary:
- Prescription-only medicine
- Toxin indicated for the temporary improvement of lines and wrinkles
- Suitable for the forehead and periorbital areas
- Free from complexing proteins
Mode of action:
- Botulinum neurotoxin type A blocks cholinergic transmission at the neuromuscular junction by inhibiting the release of acetylcholine.
- The nerve terminals of the neuromuscular junction no longer respond to nerve impulses, and secretion of the neurotransmitter at the motor endplates is prevented.
- The action results in stopping the muscular contraction of the targeted muscles.
Contraindications of Bocouture:
- Hypersensitivity to the active substance or to any of the excipients.
- Generalised disorders of muscle activity (e.g. myasthenia gravis, Lambert- Eaton syndrome).
- Infection or inflammation at the proposed injection site.
Who should not use Bocouture?
- The injection of Bocouture is not recommended for patients with a history of aspiration or dysphagia.
- Patients suffering from amyotrophic lateral sclerosis
- Patients with other diseases which result in peripheral neuromuscular dysfunction
- In targeted muscles which display pronounced weakness or atrophy
EMC: https://www.medicines.org.uk/emc/product/7418/smpc
Patient Information Leaflet: https://www.medicines.org.uk/emc/files/pil.7418.pdf
*This item requires a prescription from a registered UK prescriber before it can be dispatched*